Report: RFK Jr.’s anti-vaccine agenda curbed as GOP realizes it’s unpopular
Kennedy’s plans were only getting started. The staunch anti-vaccine activist and conspiracy theorist made his most brazen attack on vaccines in January, slashing the CDC’s childhood vaccine schedule from 17 immunizations down to 11 to be in line with recommendations of Denmark, a much smaller country with a relatively homogenous population and universal health care. The US is now an outlier among peer nations for recommending so few childhood vaccines.
Conspiracy theories and political risks
While these and other changes to vaccine recommendations by Kennedy and his underlings have been widely decried by medical and public health experts, they are still not enough for his rabid anti-vaccine followers, who, in no uncertain terms, want all vaccines abolished.
On Monday, the MAHA Institute, a think tank stemming from Kennedy’s Make America Health Again movement, held an event brimming with prominent anti-vaccine activists. Those include Del Bigtree, a prominent conspiracy theorist who leads the anti-vaccine group Informed Consent Action Network, and Mary Holland, who is CEO of the anti-vaccine group Children’s Health Defense, which Kennedy founded.
The event was focused on an alleged “Massive Epidemic of Vaccine Injury,” a nonexistent health crisis the MAHA institute wants to sell to the American public, branded as the catchy term “Mevi.” The six-hour event was essentially an extravaganza of anti-vaccine talking points, with false claims, misinformation, and disinformation about immunizations, including that vaccines cause autism and autoimmune diseases and COVID-19 vaccines are deadly.
At the start of the event, MAHA Institute President Mark Gordon laid out his grand belief that the medical community has orchestrated an elaborate, global, decades-long conspiracy to hide the dangers of vaccines, which he called poisons, and falsify data showing their benefits. “Vaccines are the greatest scam in medical history,” one of his slides proclaimed.
He concluded that “the childhood vaccination schedule needs to be eliminated and all vaccines need to be removed from the market.”
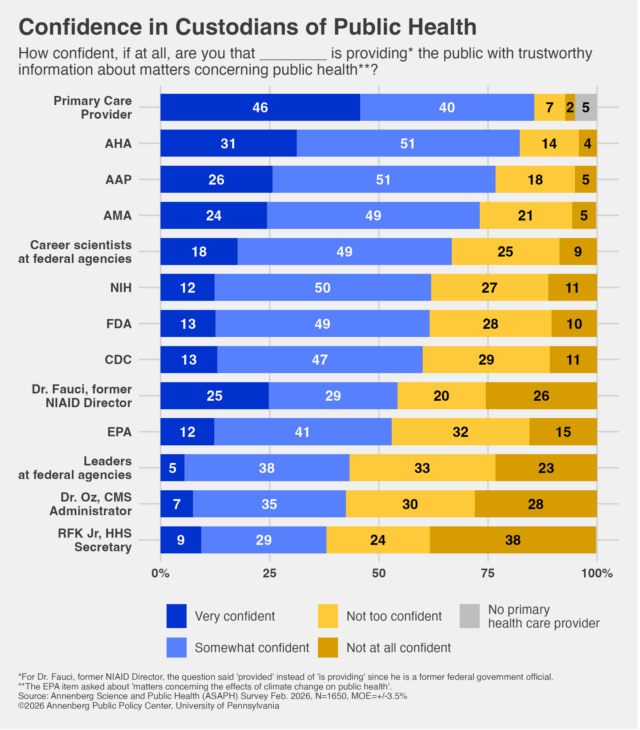
While Gordon and the other speakers were not concerned about the popularity or political ramifications of their beliefs, the Trump administration appears to be. The Post noted that Trump’s top pollster, Tony Fabrizio, has concluded that vaccine skepticism is “rejected by most voters,” and skepticism of vaccine requirements is “politically risky.” His polling data, like many others, have found broad support for vaccines and vaccine requirements. Fabrizio warned in a December memo that politicians supporting eliminating vaccine recommendations “will pay a price in the election.”
Report: RFK Jr.’s anti-vaccine agenda curbed as GOP realizes it’s unpopular Read More »